The Evolution of Our Technology
What began as intense scientific curiosity soon revealed a medical breakthrough. In 2006, Dr. Gaetano Scuderi – a world-renowned orthopedic surgeon with over 40 peer-reviewed publications in regenerative medicine – set out to answer a deceptively simple question: Why do some people develop debilitating arthritis, while others remain unaffected into old age?
The answer lay in the body’s natural defense system.
Dr. Scuderi hypothesized that variations in protease inhibitor levels might explain why some joints degrade rapidly while others are protected. He knew from surgical experience that three specific protease enzymes were primarily responsible for cartilage destruction in osteoarthritis. Scouring the medical literature, he identified a powerful and overlooked molecule in the blood: Alpha-2-Macroglobulin (A2M) — a broad-spectrum protease inhibitor with the potential to act as the body’s “guardian” against joint degeneration. From this insight, Cytonics was born – with a mission to harness A2M’s natural power and build next-generation therapies that could change the trajectory of osteoarthritis for millions.
Some patients worsened after spine surgery without signs of infection, prompting Dr. Scuderi to investigate this mystery by identifying the biological mechanism behind the phenomenon.
Dr. Scuderi theorized that a protein family called proteases were the cultprit. After injury, proteases typically clear damaged tissue. But in some cases, they get stuck in the "on" position and remain overactive past their usefulness - damaging cartilage in the long run.
Dr. Scuderi's hunch was confirmed by his discovery of the Fibronectin Aggrecan Complex (FAC) - a biomarker ofprotease mediated cartilage degeneration - pointing to proteases as the root cause of chronic cartilage damage.
He hypothesized that protease-driven inflammation causes a vicious cycle of cartilage degradation. A broad-spectrum protease inhibitor might be the key to interrupting it.
The search was on. Dr. Scuderi scoured the literature and discovered a natural protease inhibitor found in the blood: Alpha-2-Macroglobulin (A2M), a natural protease inhibitor that plays a role in blood clotting - could A2M be repurposed to treat joint damage?
Cytonics' first-gen APIC therapy uses a patient’s own A2M protein to treat arthritis. It’s helped over 10,000 people and validated A2M’s potential, but the process is slow, inconsistent across patients, and difficult to scale for broader use.
Using next-generation genetic engineering, the Cytonics team develops CYT 108 - a synthetic version of A2M that’s 200% more potent and optimized to potently inhibit the multiple proteases that cause osteoarthritis.
After 7 years and over $10M in development, CYT-108 enters Phase 1 trials in June 2024. Early data shows it’s safe, paving the way for larger efficacy studies.
From Proof To Platform: Harnessing the Power of A2M
The First Generation Technology
Much has been made of “regenerative orthopedics”. Stem cells, PRP, and other autologous treatments promising cartilage repair. But most of these therapies are unproven, unregulated, and un-reimbursable. That’s where Cytonics broke away from the pack.
Our first-generation therapy, APIC™ (the Autologous Protease Inhibitor Concentrate), put Alpha-2-Macroglobulin (A2M) on the clinical map and into the offices of orthopedists nationwide.
A2M is a naturally occurring molecule found in your blood – a broad-spectrum protease inhibitor that binds to and neutralizes the enzymes responsible for breaking down cartilage in osteoarthritis. Using a proprietary filtration process, we developed APIC™ to isolate and concentrate A2M from a patient’s own blood, then re-inject it directly into the affected joint to halt cartilage degeneration and promote regeneration at the source.
The results were undeniable: Over 10,000 patients treated. 300+ physicians trained. Strong clinical and commercial validation.
APIC™ became the first and only autologous A2M treatment to receive FDA IND approval — a critical milestone toward formal marketing authorization and future reimbursement. But as effective as APIC™ is, it has limitations: variable A2M levels between patients, multi-hour processing times, and specialized equipment that restricts scalability.
These constraints inspired the next evolution in our platform – a synthetic, highly potent version of A2M designed for global scale and wide-spread accessibility: CYT‑108.


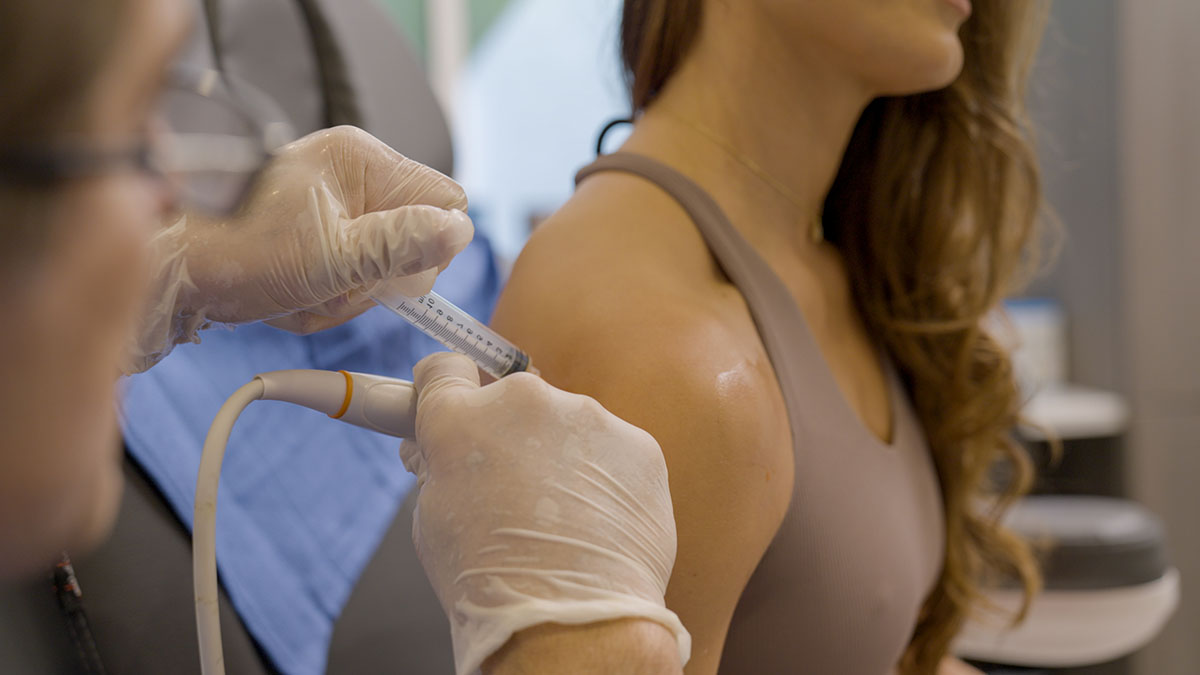

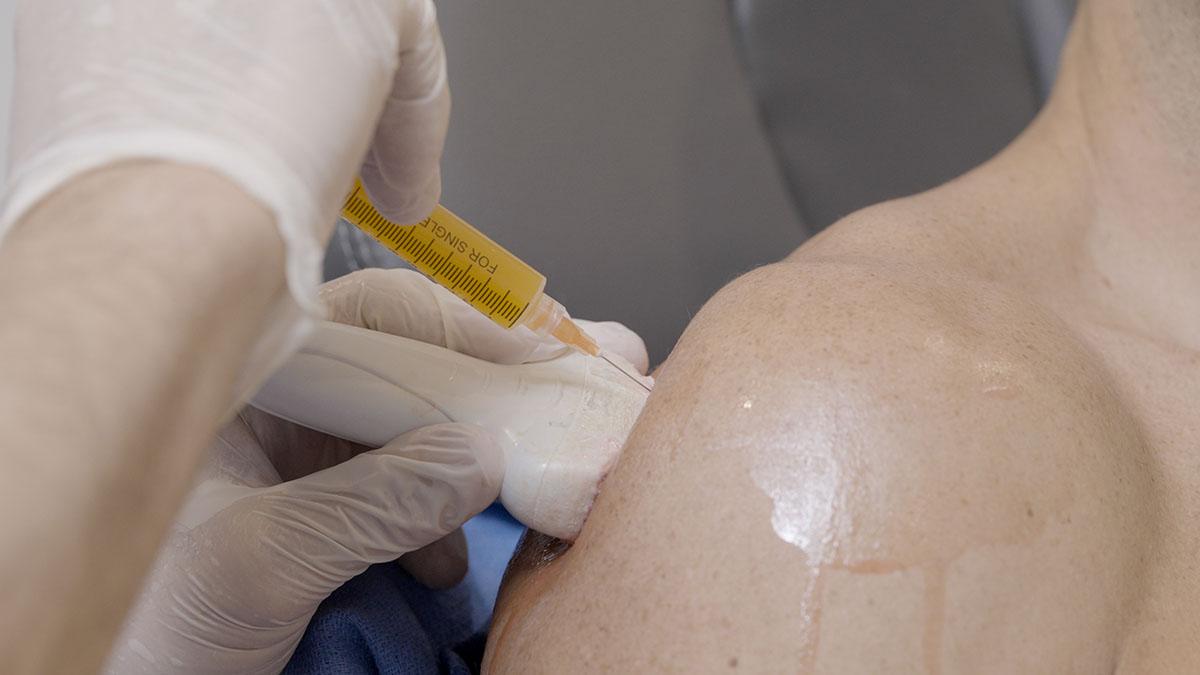


The Next Generation: CYT-108
Our next-generation therapy, CYT‑108, is a synthetic, precision-engineered version of Alpha-2-Macroglobulin (A2M) – built for superior potency, purity, and consistency in treating osteoarthritis.
Unlike traditional therapies that merely mask symptoms, CYT‑108 targets the root cause: overactive proteases that destroy cartilage. It blocks all three major enzyme families – MMPs, ADAMTSs, and Cathepsins – making it both broad-spectrum and highly specific. Think of it as a molecular Swiss Army knife, engineered to dismantle the biochemical machinery of OA.
But CYT‑108 does more than stop destruction – it may help rebuild what’s lost. Independent preclinical research has shown that A2M promotes chondrocyte proliferation and cartilage matrix synthesis, including increases in type II collagen and aggrecan – the two molecules responsible for cartilage structure and resilience. These effects were driven by A2M’s upregulation of PCNA, a key protein involved in cellular regeneration (Guo et al., Cartilage, 2025).
Administered via a targeted intra-articular injection, CYT‑108 is delivered directly into the joint space, ensuring maximum impact at the site of disease while minimizing systemic exposure. This precision reduces side effects and enhances therapeutic efficiency – a major leap forward in OA treatment.
What Makes CYT‑108 Better Than Natural A2M?
Cytonics has genetically engineered a “super A2M” designed to specifically target and efficiently eliminate ALL major classes proteases involved in OA.
Our first-generation therapy APIC™ proved that the natural A2M protein is an effective therapy for osteoarthritis, capable of targeting the root molecular causes of the disease instead of merely masking symptoms. But despite its clinical and commercial success, APIC™ has practical limitations: concentrating A2M from patients’ blood is a time-consuming processing, there are variable A2M levels (and therefore variable potency) between patients, and logistical challenges make it difficult to scale for widespread adoption.
CYT‑108 changes everything. It’s a lab-manufactured, precision-engineered version of A2M designed to overcome the bottlenecks of autologous therapy and unlock the full potential of A2M’s protease inhibition and cartilage regeneration activities.
Because CYT‑108 is shelf-stable and produced in controlled conditions, it offers precise dosing, 200% greater potency than natural A2M, and the potential for global manufacturing and distribution. For the first time, a scalable A2M therapy is within reach.
Unlike Big Pharma’s failed attempts to target individual protease enzymes, CYT‑108 is a “Super A2M” that neutralizes all major proteases responsible for cartilage breakdown in OA. That means broad-spectrum efficacy, more consistent results, and greater impact on long-term patient well-being.
Natural A2M:
APIC purified from patient blood
CYT-108:
genetically engineered (recombinant) A2M
| APIC™ | CYT-108 | |
|---|---|---|
| Source | Patient’s own blood | Genetically-engineered (recombinant A2M) |
| Efficacy | Patient-to-patient variability | Precisely dosed, 200% more potent & optimized against MMP13 and ADAMTS-5 |
| Scalability | Limited by blood processing | Laboratory production |
| Insurance Coverage | Uncertain | High potential |
How CYT-108 Works
A2M is a naturally-occurring protease inhibitor that protects against cartilage degradation. Though levels are too low in joints to be an effective treatment…
CYT-108 is a genetically engineered “super” A2M” protein that is 200% more potent than the naturally-occurring A2M.
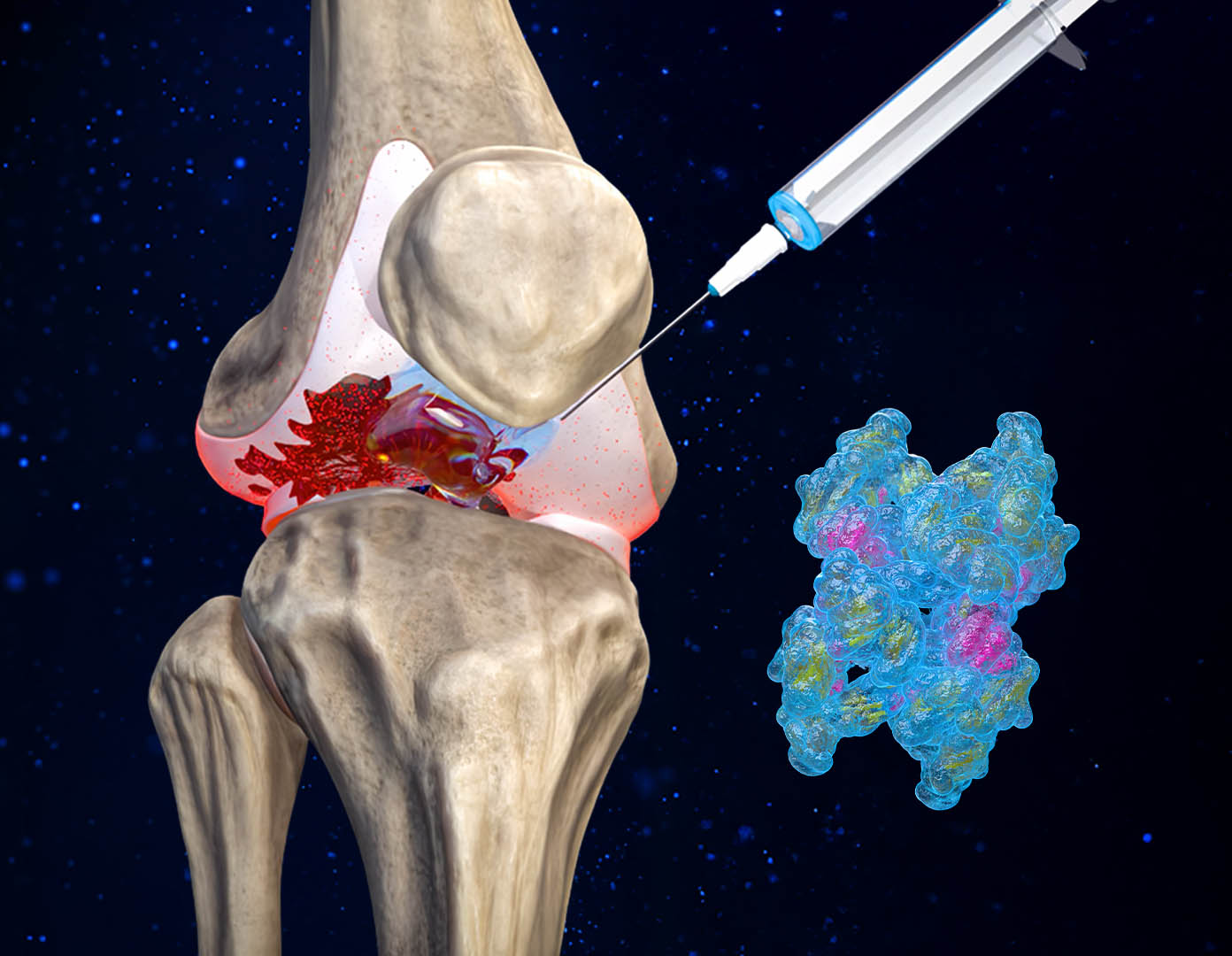
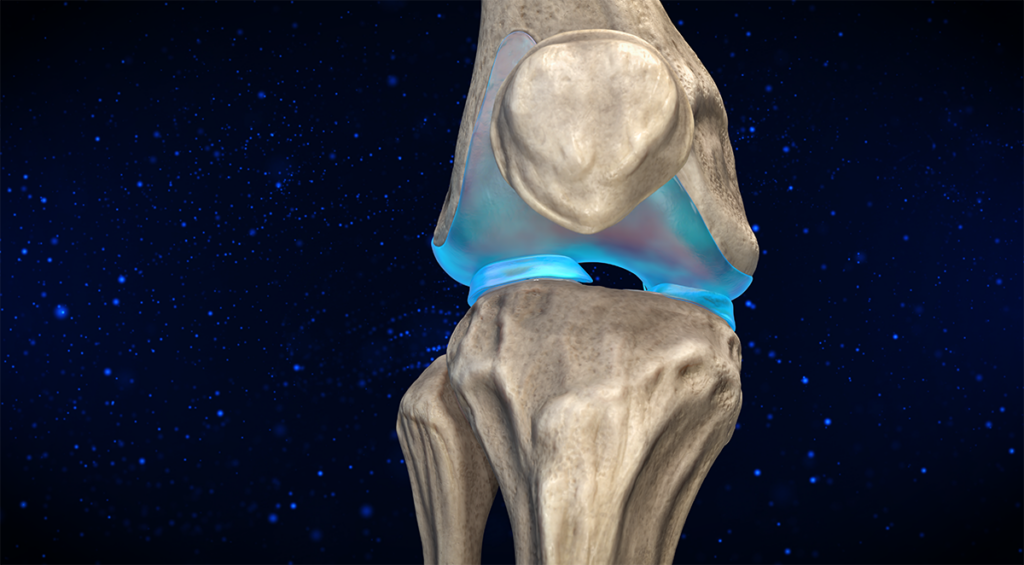
Intra-articular (targeted) delivery for maximum concentration and minimum off-target effects
1.
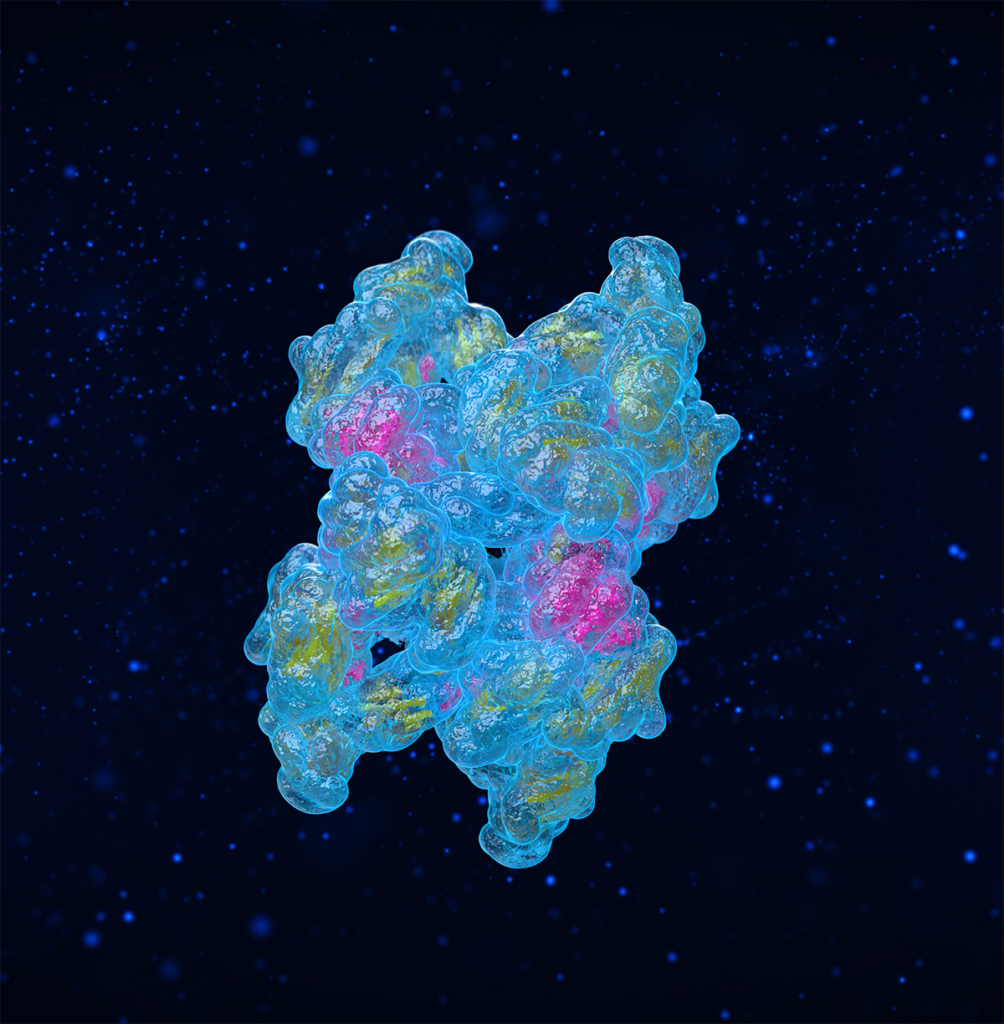
Sequesters cytokines and neutralizes destructive proteases, cutting off the inflammatory feedback loop
2.

Designed to quickly reduce joint pain and inflammation while halting progressive cartilage degradation
3.
Fibronectin-Aggrecan Complex Test (FACT™)
Through the discovery of a unique “pain protein” that can identify the source of musculoskeletal pain, Cytonics developed the Fibronectin-Aggrecan Complex Test (FACT™), a diagnostic molecular biomarker test to identify symptomatic intervertebral discs or painful joint abnormalities. FACT™ is used to measure the presence of the Fibronectin-Aggrecan Complex (FAC) in a fluid specimen taken from patients with joint or disc pain. The FAC is a unique molecular complex that has been reported in peer-reviewed publications to be associated with painful inflammation.
FACT™ is a novel antibody test that was developed as an optimized assay to detect the Fibronectin-Aggrecan Complex in samples of patients’ joint fluid. The test is a heterogeneous sandwich ELISA utilizing anti-fibronectin antibody immobilized on solid support and anti-aggrecan-G3 as a detection antibody. It can be used by physicians for testing synovial fluid from any joint or from intradiscal fluid obtained by Molecular Discography. Your FACT™ score can help your physician determine what is going on below the surface, beyond those signs and symptoms you and your doctor can see and feel.
Physicians commonly use MRI imaging to identify orthopedic abnormalities. However, MRI has limitations. Many patients have “normal” looking anatomy following a traumatic injury even though degenerative enzymes may be at work causing pain and slowly destroying their joint. Additionally, patients often have non-painful abnormalities that are indistinguishable from painful conditions on an MRI image. The limitations of MRI often lead to misdiagnosis and ineffective treatment, leaving patients to continue to suffer from joint pain following treatment despite the best intentions of their physicians.